Time:2026-02-26
Researchers led by CAS member YAN Jun from the Institute of Neuroscience, Center for Excellence in Brain Science and Intelligence Technology , Chinese Academy of Sciences, in collaboration with the School of Basic Medical Sciences and Zhongshan Hospital, Fudan University. The research published on Neuron reconstructed the cellular architecture and spatial transcriptomic atlas of the human suprachiasmatic nucleus (SCN)—the core brain region regulating circadian behavior—at the single-cell level, and systematically dissected the neuronal subtypes of the human SCN.
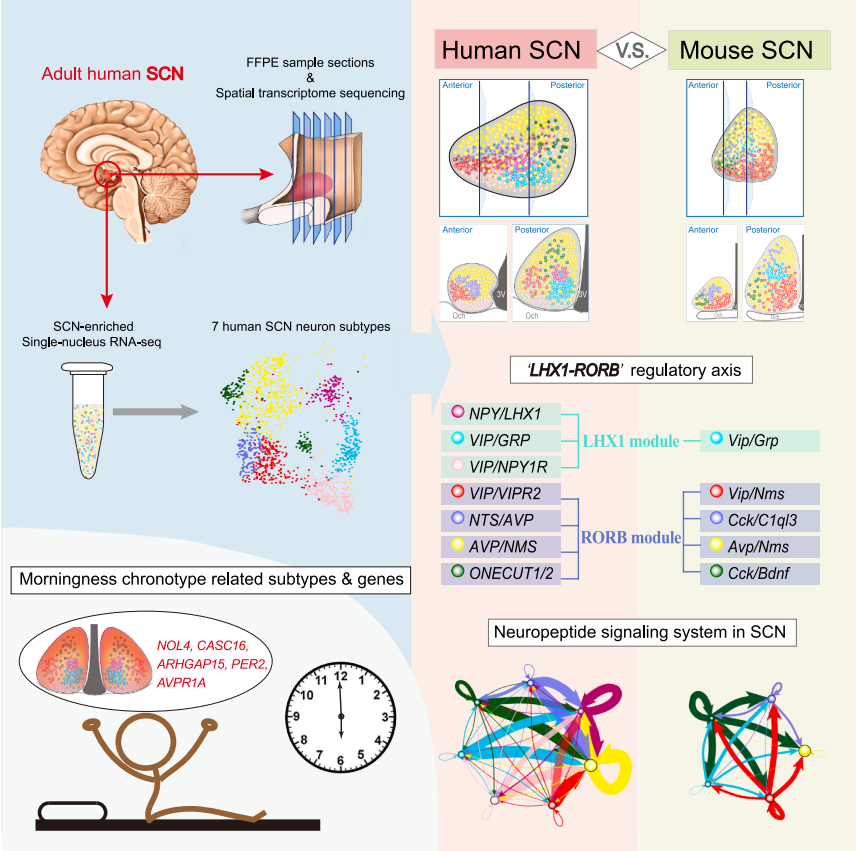
Figure 1: Graphic summary of the main findings of this study
Circadian rhythms are endogenous timing mechanisms evolved by organisms to adapt to the Earth's 24-hour periodic environmental changes, playing a critical role in regulating animal behaviors (such as sleep-wake cycles) and physiological processes (such as body temperature regulation). Circadian disruption can lead to various health issues, including sleep disorders, mental illness, metabolic abnormalities, cancer, and cardiovascular and cerebrovascular diseases, with a significant impact on human health and social development.
At the molecular level, circadian rhythms are maintained by transcriptional-translational feedback loops composed of a set of core clock genes. Research on the molecular mechanisms of circadian rhythms was awarded the 2017 Nobel Prize in Physiology or Medicine. Although almost all cells possess individual circadian oscillators, neurons related to circadian rhythm in the brain regulate circadian behaviors through complex neural networks.
The mammalian master circadian clock is located in the suprachiasmatic nucleus of the hypothalamus, responsible for orchestrating rhythms across other brain regions and peripheral tissues. Reconstructing and deciphering the fine structure and function of the circadian clock network in the brain is key to understanding the neural mechanisms of rhythmic behavior.
To explore these questions, the YAN Jun research group at the Center for Excellence in Brain Science and Intelligence Technology had previously mapped the spatiotemporal single-cell transcriptomic atlas of the mouse SCN, completed structural reconstruction and neuronal subtype identification (Wen et al., 2020), and discovered a novel subtype of SCN neurons—the CCK neuron. Further functional studies revealed that CCK neurons play a critical role in maintaining the robustness of circadian rhythm (Xie et al., 2023).
However, these studies were conducted only in rodents (mice), and these results cannot be directly applied to the study of the human circadian clock. Furthermore, human populations exhibit different temporal preferences in daily rhythmic behavior, termed chronotypes, such as "morningness" and "eveningness." Although genome-wide association studies (GWAS) have identified numerous candidate genes associated with human chronotypes, whether and how these genes affect the function of the human SCN remains unclear. Studies on the structure and function of the human SCN will help advance the clinical investigation of circadian rhythm-related diseases.
In this study, the researchers combined spatial transcriptomics, single-nucleus RNA sequencing technology, and deep learning-based histological analysis to reconstruct a human SCN cellular atlas, integrating histological information, cellular composition, and gene expression profiles. Through a comprehensive three-dimensional reconstruction of the human SCN, they discovered that it is enriched with vascular networks and connected to the organum vasculosum of the lamina terminalis (OVLT) via these vessels, suggesting that human SCN might control rhythmic activities in other brain regions through this portal vein system.
The study further identified seven human SCN neuronal subtypes with specific transcriptomic profiles and spatial distributions. Comparison with SCN transcriptomes from mice and non-human primates (macaques) revealed a high degree of cross-species conservation in SCN gene modules and functional segregation regulated by the transcription factors LHX1 and RORB. Furthermore, cross-species comparative analysis indicated significant reorganization of the neuropeptide signaling network in the human SCN.
Finally, by mapping GWAS-related genes onto the human SCN atlas, the researchers found that the AVP/NMS neuronal subtype was enriched for genes associated with the human morningness chronotype, providing a potential explanation for the neural mechanisms underlying chronotype differences in populations.
In summary, this study has, for the first time, mapped a comprehensive cellular atlas of the human SCN, revealing neuropeptide signaling pathways unique to the human SCN and gene regulatory programs conserved across mammalian species, and identified potential neural mechanisms underlying chronotype differences in human populations.
Contact:
YAN Jun
Institute of Neuroscience, Center for Excellence in Brain Science and Intelligence Technology, Chinese Academy of Sciences, Shanghai, China.
junyan@ion.ac.cn
 附件下载:
附件下载: